In previous post, we mentioned that enantiomers (also known as optical isomers) rotate plane-polarised light in opposite directions and are said to exhibit optical activity. So how do we know if an organic molecule rotate plane-polarised light? We use an instrument known as a polarimeter.
A polarimeter is a scientific instrument used to measure the angle of rotation caused by passing polarised light through an optically active compound.
Some chemical substances are optically active, and polarized (uni-directional) light will rotate either to the left (counter-clockwise) or right (clockwise) when passed through these compounds. The amount by which the light is rotated is known as the angle of rotation.
Today, we shall take a closer look at:
- Meaning of Optical Activity
- Plane-Polarised Light
- How a Polarimeter Works
Meaning of Optical Activity
All molecules, whether chiral or achiral, are able to rotate plane-polarised light due to interaction with the electron cloud of the molecule. For a particular molecule, the extent and direction of rotation depends on the orientation of the molecule in relation to the plane-polarised light.
For achiral molecules (compounds not exhibiting enantiomerism), each of these rotations are cancelled out due to the symmetry of the molecules, and thus there’s no net rotation of the plane-polarised light. Hence, there’s no optical activity.
For chiral molecules (compounds exhibiting enantiomerism), such exact cancellation of individual rotation is not possible as the chiral molecule are not symmetrical. This results in optical activity i.e. rotation of the plane-polarised light, and it can be measured with a polarimeter.
Do note that in a racemic mixture (also known as racemate), where there are equal amounts of both enantiomers, the equal and opposite rotation of plane-polarised light cancels out, and results in optical inactivity also. In contrast to the two enantiomers, which have identical physical properties except for the direction of rotation of plane-polarised light, a racemic mixture sometimes has different properties from either form of the pure enantiomers. Examples could be different in melting points, boiling points and solubilities.
Plane-Polarised Light
Typical light sources are non-polarised. This means that the light consists of light of all polarisations. By passing the light through a polariser, only light of one particular polarisation is allowed to pass through. The light which has passed through the polariser is known as plane-polarised light.
How a Polarimeter works
What we see normally is unpolarised light, which tends to vibrate randomly in all directions. Plane-polarised light is composed of waves that vibrate in only one plane. When polarised light passes through a polariser, the randomly vibrating light waves are filtered so that the light passing through is vibrating in only one direction.
Below is a schematic diagram of a polarimeter.
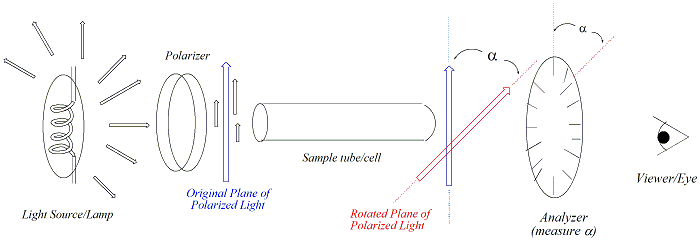
A polarimeter is used to measure the rotation of polarised light. Monochromatic (one colour) light from the light source passes through a polariser, and then through the sample cell containing a solution of an optically active compound. On leaving the sample cell, the polarised light encounters another polariser, better known as an analyser. This analyser is movable and the operator rotates it until the maximum amount of light is transmitted, then reads the angle of observed rotation.
I hope you find the content easy for your understanding and if you have any questions, leave me a comment below. Feel free to share this blog post with your friends.
Subscribe to my blog to receive 2 updates per month sent to your email!
PS: Under related articles below, there are several blog post discussions and questions related to Isomerism in Organic Chemistry. You can also do a keyword search using the search box at the top right hand corner.
Related Articles:
- Organic Chemistry: Meso Compounds and Maximum Possible Numbers of Enantiomers
- Organic Chemistry: Optical Isomerism
- Organic Chemistry: Special (Unusual) Cases of Chirality and Achirality
- Organic Chemistry: Total Number of Stereoisomers
- Organic Chemistry: Key Concepts in Introductory Topics, Alkanes, Alkenes & Arenes

Leave a Reply