Chirality of Molecule
First and foremost, let’s recap on the meaning of chirality in an organic molecule.
We have learned that there must be at least one chiral centre in the molecule for it to be chiral and exhibits enantiomerism (optical isomerism). Chiral centre is a tetrahedral atom (usually carbon) that has four different substituent groups on it. This gives rise to enantiomers. Due to the chiral centre, there is an absence of a internal plane of symmetry in the molecule. A internal plane of symmetry is any plane cutting through the molecule such that one side is a perfect reflection of the other. For molecules with an internal plane of symmetry, we say that they are achiral and will not exhibit enantiomerism.
Three quick questions for you to think through before we move on:
- Must a molecule with chiral centre(s) be always chiral?
- Must a molecule without a plane of symmetry be always chiral?
- Must a molecule have a chiral centre in order to be chiral?
There are 3 special (unusual) cases of chirality and achirality which JC students taking A-Level H2 Chemistry syllabus should take note, namely:
- Meso compounds
- Some molecules without a plane of symmetry can also be achiral
- Chiral molecules without any chiral centres
1. Meso Compounds
Meso compounds are achiral molecules which possess multiple chiral centres (i.e. more than one chiral centres). They have an internal plane of symmetry which cause their mirror images to be superimposable. This results in them being achiral i.e. no optical activity. You can check out a previous blog post dedicated solely to meso compounds.
2. Some Molecules Without A Plane of Symmetry Can Also Be Achiral
We have learned that while a molecule with an internal plane of symmetry must be achiral, some molecules without an internal plane of symmetry can also be achiral.
Let’s take molecule W for an example for discussion. It has no internal plane of symmetry, yet its mirror image is superimposable on itself. As such, molecule W is achiral.
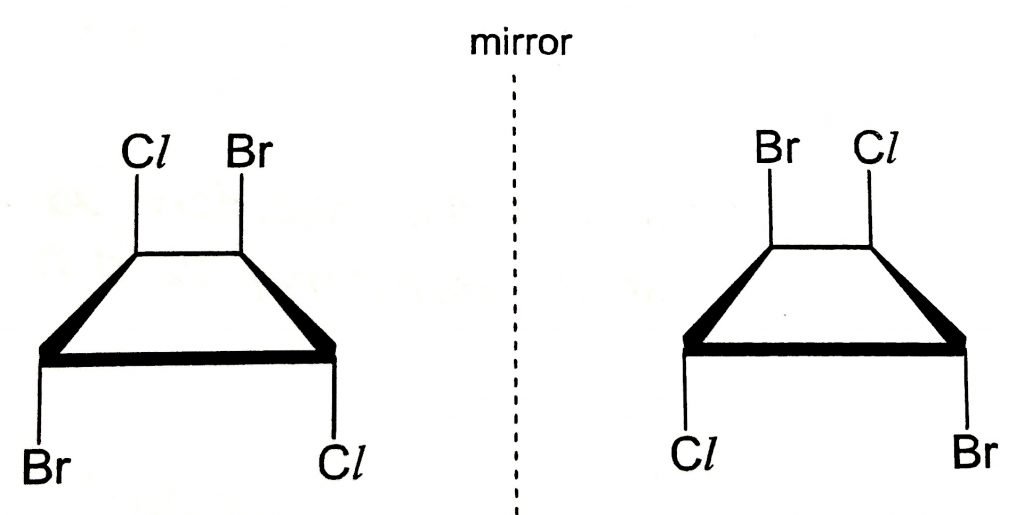
It is important to have some visualisation skills to help you determine if such molecule is chiral or achiral. You can flip the mirror image of molecule W about the plane of the ring and rotate it 180o to superimpose it with the molecule on the left-hand side.
3. Chiral Molecules Without Any Chiral Centres
Enantiomers can exists in compounds even without any chiral centres. This happens in some allenes.
An allene is a compound in which one carbon atom has C=C double bonds with each of its two adjacent carbon centres.
The simplest known allene is propadiene, H2C=C=CH2 which is achiral. This is because the molecule has an internal plane of symmetry. The mirror image of propadiene is superimposable with it.

Recall that in order for a molecule to be chiral it must not be superimposable on its mirror image. If the molecule has a mirror plane (internal plane of symmetry) then it will be superimposable on its mirror image [and therefore achiral].
So when will an allene exhibits chirality?
When the two outer doubly bound carbons each have two different groups attached then the molecule will lack an internal plane of symmetry and is thus chiral.
Consider the molecule 2,3-pentadiene, CH3CH=C=CHCH3:
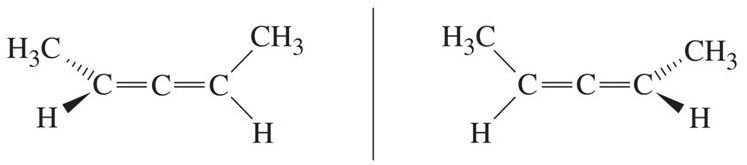
Due to restriction of rotation about the C=C double bonds, the two terminal methyl groups lie in the perpendicular planes, and there is no internal plane of symmetry in the molecule.
As such, 2,3-pentadiene molecule is chiral and there are two non-superimposable enantiomers which exist.
So far so good? How about testing yourself on identifying chiral and achiral allenes?
Test Yourself
Question:
Which of the following allenes (molecules A-D) are chiral, and which are achiral?
Do write down your answers in the Comments Section below.




I hope you find the content easy for your understanding and if you have any questions, leave me a comment below. Feel free to share this blog post with your friends.
Subscribe to my blog to receive 2 updates per month sent to your email!
PS: Under related articles below, there are several blog post discussions and questions related to Isomerism in Organic Chemistry. You can also do a keyword search using the search box at the top right hand corner.
PPS: If you need help with your A-Level H2 Chemistry, do join us in our weekly A-Level H2 Chemistry Tuition Class.
