First of all, re-cap that isomerism can be broadly classified into 2 types:
- Constitutional isomerism, in which atoms are linked together in different ways
- Stereoisomerism, in which atoms have the same connectivity but different arrangements in space.
Today, we will be talking about constitutional isomerism.
Constitutional isomers have the same molecular formula but different structural formulae. This simply means that the atoms are linked together in different ways. They are also known as structural isomers.
There are three types of constitutional isomerism:
- Positional Isomerism
- Chain Isomerism
- Functional Group Isomerism
1. Positional Isomerism
Positional isomers are molecules with the same molecular formula and same functional group but differ in the position of the functional group.
Positional isomers are also commonly known as compounds having the same substituents at different positions on the same carbon skeleton.
Positional isomers have similar chemical properties but different physical properties.
Examples of positional isomers:



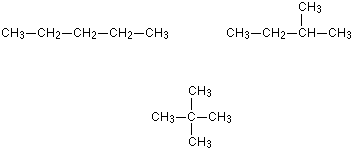
2. Chain Isomerism
Chain isomers refers to organic compounds with the same molecular formula and same functional group, but different carbon skeleton.
Chain isomers have similar chemical properties but different physical properties.
Examples of chain isomers:
3. Functional Group Isomerism
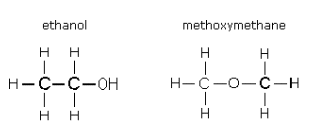
Functional group isomers are organic molecules that have the same molecular formula but different functional groups.
Each isomer share the same molecular formula but contains a different functional group other than the hydrocarbon skeleton.
Functional group isomers have very different physical and chemical properties.
Examples of functional groups isomers:

I hope you find the above discussion useful towards your learning of Isomerism in Organic Chemistry.
Before we end this post, how about trying out a H2 Chemistry exam question on constitutional isomerism? Do leave your answer below in the Comments Section below.
Question:
How many constitutional isomers are there in C3H6Br2?
I hope you find the content easy for your understanding and if you have any questions, leave me a comment below. Feel free to share this blog post with your friends.
Subscribe to my blog to receive 2 updates per month sent to your email!
PS: Under related articles below, there are several blog post discussions and questions related to Isomerism in Organic Chemistry. You can also do a keyword search using the search box at the top right hand corner.
PPS: If you need help with your A-Level H2 Chemistry, do join us in our weekly A-Level H2 Chemistry Tuition Class.
