
Today, we are going to discuss a very important concept in Atomic Structure and Chemical Bonding, known as Ionisation Energy.
We will start by looking at First Ionisation Energy, 1st IE which allows you to deduce the Second Ionisation Energy, 2nd IE and more.
You will also understand how to determine the maximum number of ionisation energies of an element in the Periodic Table.
First Ionisation Energy, 1st IE
It is defined as the amount of energy required to remove 1 mole of electrons from 1 mole of gaseous atoms to form 1 mole of singly charged gaseous cations.
It can be represented by the following equation: X (g) → X+ (g) + e– ∆H1 = 1st ionisation energy = +ve
Second Ionisation Energy, 2nd IE
It is defined as the amount of energy required to remove 1 mole of electrons from 1 mole of singly charged gaseous cations to form 1 mole of doubly charged gaseous cations.
It can be represented by the following equation: X+ g) → X2+ (g) + e– ∆H2 = 2nd ionisation energy = +ve
All the ionisation energies are positive values since energy is absorbed in removing an electron from an atom or ion.
Determination of the Maximum Number of Ionisation Energies of an Element
The number of ionisation energies that an element can have is always equal to the atomic number (proton number).
For example, oxygen has atomic number of 8 and thus the maximum ionisation energy of oxygen is until 8th Ionisation Energy, 8th IE.
Let’s take a look at the following YouTube Video for more examples on Ionisation Energy.
Length of video: 6.17 minutes
Direct video link: https://youtu.be/TJMSTNoR7lQ
From the video above, we have discussed about the formal definition of 1st IE and as well other ionisation energies. We have also learned how to determine the maximum number of ionisation energies that are possible for an element.
3 important points to take note for Ionisation Energy:
- Ionisation energies normally have positive values since energy is absorbed in removing an electron
- Successive ionisation energies of an element increase with the removal of each electron because the remaining electrons are attracted more strongly by the constant positive charge on the nucleus
- The number of ionisation energies that an element can have equals its atomic/proton number
For students taking the Singapore Cambridge GCE A-Level H2 Chemistry syllabus, you can observe the above 3 points in your Chemistry Data Booklet given by your JC Chemistry Tutors or our JC A-Level H2 Chemistry Tuition Classes. You can also download them at Singapore Examination & Assessment Board (SEAB) website. A sample page of ionisation energy is shown below.
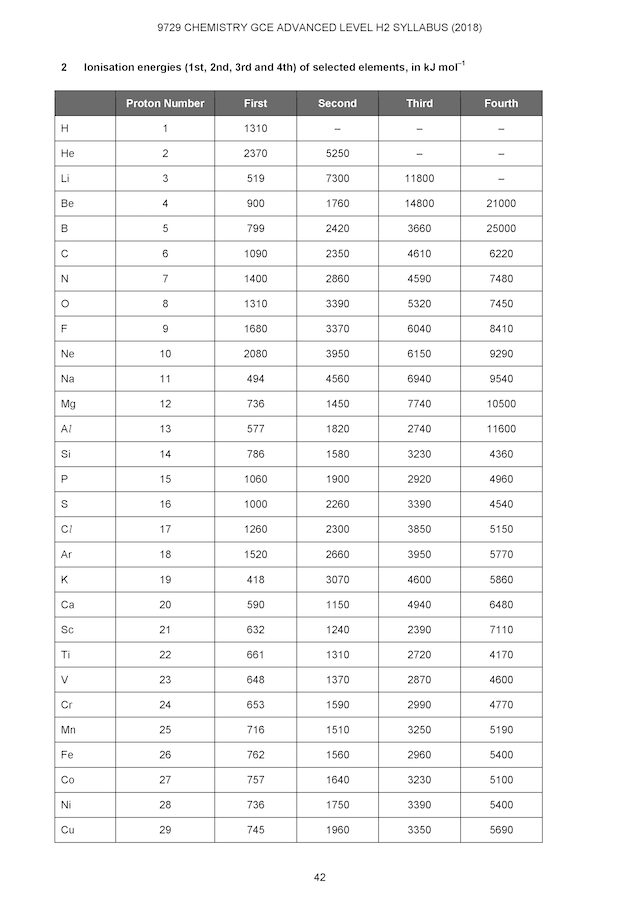
Now, how about testing yourself on what we have just discussed?
Quick Check 1:
How many ionisation energies can Aluminium (proton number 13) have?
Quick Check 2:
Write the equation for the 7th ionisation energy for Oxygen (proton number 8).
I hope you find the content easy for your understanding and if you have any questions, leave me a comment below. Feel free to share this blog post with your friends. This topic is usually discussed in JC1 (Junior College Year 1) in Singapore.
Subscribe to my blog to receive 2 updates per month sent to your email!
PS: Under related articles below, there are several blog post discussions and questions related to Atomic Structure and Ionisation Energies. You can also do a keyword search using the search box at the top right hand corner.
