In the previous blog posts, we have talked about the different Standard Enthalpy Changes of Reactions as well as look into using Energy Cycles with Hess’ Law to solve Chemistry questions related Chemical Energetics.
Today, i am going to re-post a question for discussion which i have blogged about several years back. This questions is evergreen and you will be seeing such questions again in your upcoming A-Level H2 Chemistry examinations.
The following question was sent by one of our A-Level JC1 H2 Chemistry students in Singapore.
It is a very is simple yet interesting MCQ question and requires the students to be very good in drawing the Energy Cycle Diagram, in order to solve the question.
In fact, you can also solve this question using Algebraic Method.
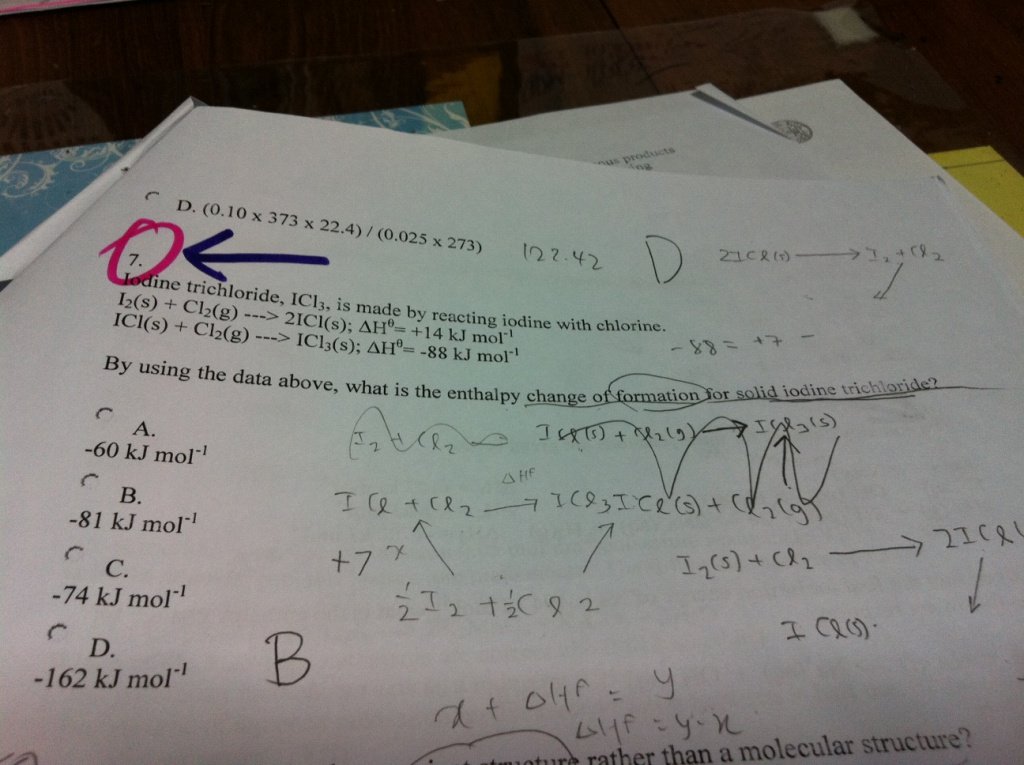
My student thinks that the answer should be option “B”‘. Do you agree with her?
Show your working or brief explanation on arriving at your answer.
“80% of the Success is about taking the Necessary Actions”
I hope you find the content easy for your understanding and if you have any questions, leave me a comment below. Feel free to share this blog post with your friends.
Do stay tuned to the upcoming posts as i will be discussing on the applications of Enthalpy Changes using Hess’ Law. I will also be sharing the concepts and applications involved in Entropy Changes and Gibbs Free Energy.
Subscribe to my blog to receive 2 updates per month sent to your email!
PS: Under related articles below, there are several blog post discussions and questions related to Chemical Energetics. You can also do a keyword search using the search box at the top right hand corner.
PPS: If you need help with your A-Level H2 Chemistry, do join us in our weekly A-Level H2 Chemistry Tuition Class.
Related Articles:
- Chemical Energetics: Application of Hess’ Law & Energy Cycle Diagram
- Chemical Energetics: Definitions of Standard Enthalpy Changes of Reactions
- Chemical Energetics: Application of Gibbs Free Energy in Thermodynamics
- Chemical Energetics: Experimental Method to Determine Enthalpy Change of Combustion
- Chemical Energetics: Entropy (S)

yeah i think its B too. release the soln pls!!!
The enthalpy of formation is represented by the equation:
1/2I2 + 3/2Cl2 –> ICl3
To get this you can multiply equation 1 by 1/2 and then add equation 2
Hf(ICl3) = +7 – 88 = -81 kJ
My answer is the same as hers. (+14/2)+(-88)=-81kJ/mol.
I2 + Cl2 (+2Cl2) —————–> 2ICl3 {1}
2ΔHf(ICl3)
I2 + Cl2 (+2Cl2) —————> 2ICl + 2Cl2 {2}
+14 kJmol^-1
2ICl +2Cl2 ——————–> 2ICl3 {3}
2(-88) kJmol^-1
It’s difficult to show the trianglular diagram here, so just try to visualize the triangle. Ignored the state symbols to avoid getting messy. Won’t be doing that in the real exams though.
In my working, I’d put eqn {1} on top, and {2} and {3} in the left and right bottom corners of the diagram respectively.
You can see that the (+2Cl2) in {1}will sort of “balance” eachother out, so there’s no need to consider bond energy of Cl2.
By Hess’ Law, 2ΔHf(ICl3)= 14+2(-88)= -162 kJmol^-1
∴ ΔHf(ICl3)= -81 kJmol^-1
So I guess the girl would have been correct, answer B.
Let me know if you spot any errors, thanks.
Yes,I’m agree with her asnwer~
Firstly, we will get an answer (-162 kJ per mole) for 2 iodine trichloride~ since enthalpy change of formation is meant for 1 mole , we have to divide -162/2 and thus we will get -81 kJ mol-1 as our answer~ Is my concept correct, Sir? thanks for guiding me~