Most of the daily products we encounter contain dissolved chemicals which can be harmful when present in high concentrations. For example, too much alkali in body wash and shampoos can cause skin irritation. Chemists can check the concentration of these chemicals by carrying out volumetric analysis.
To do volumetric analysis, we use a method called titration which involves:
- a solution of unknown concentration (analyte in conical flask)
- a solution of known concentration (titrant in the burette)
Titrations are used to obtain quantitative information about chemical reactions, such as the concentration of a solution or establish the stoichiometry of a reaction.
Acid-Base Titrations
Direct titrations that involve the use of an acid, such as hydrochloric acid and a base, such as sodium hydroxide, are called acid-base titrations.
In a typical titration, a known volume of a standard solution of one reactant (or a reactant with known concentration) is measured into a conical flask, using pipette. A solution of the other reactant (with unknown concentration) is then added, from a burette, slowly into the conical flask, until the reaction between the two substances is complete.
For an acid-base neutralisation reaction, the completion of the reaction is often found by noting the colour change of an indicator added to the reaction flask.
This allows us to determine the volume of titrant required to react completely with a fixed volume of a solution of unknown concentration (analyte).
The unknown concentration can then be easily determined by performing stoichiometric calculations.
Back Titrations
So, when do we need back titrations?
Back titrations are usually used when a direct titration is not possible.
e.g. when it involves solid substances such as CaCO3, where the end-point is difficult to detect.
e.g. when it involves volatile substances such as ammonia and iodine, where inaccuracy arises due to loss of substance during titration.
In back titration, a known excess of one reagent A is allowed to react with an unknown amount of B. The amount of unreacted A is then determined by titration with a reagent C of known concentration. From the titration results, the amount of unreacted A and the amount of B can be found by simple stoichiometric calculations.
5 Simple Steps in Back Titration Calculations:
- Determine the amount of C required in the titration
- Using stoichiometry, find the amount of A that reacted with C in the titration
- Note that amount of A that reacted with C in the titration = amount of A that did not react with B in the earlier reaction
- Therefore, amount of A that reacted with B in the earlier reaction = total amount (known excess) of A added initially – amount of A that did not react with B earlier (i.e. amount of A that reacted with C in the titration)
- Knowing now, the amount of A that reacted with B, using stoichiometry, the amount of B can be found
The following bar model will help you to understand the how the 5 steps are being used to solve back titration questions.
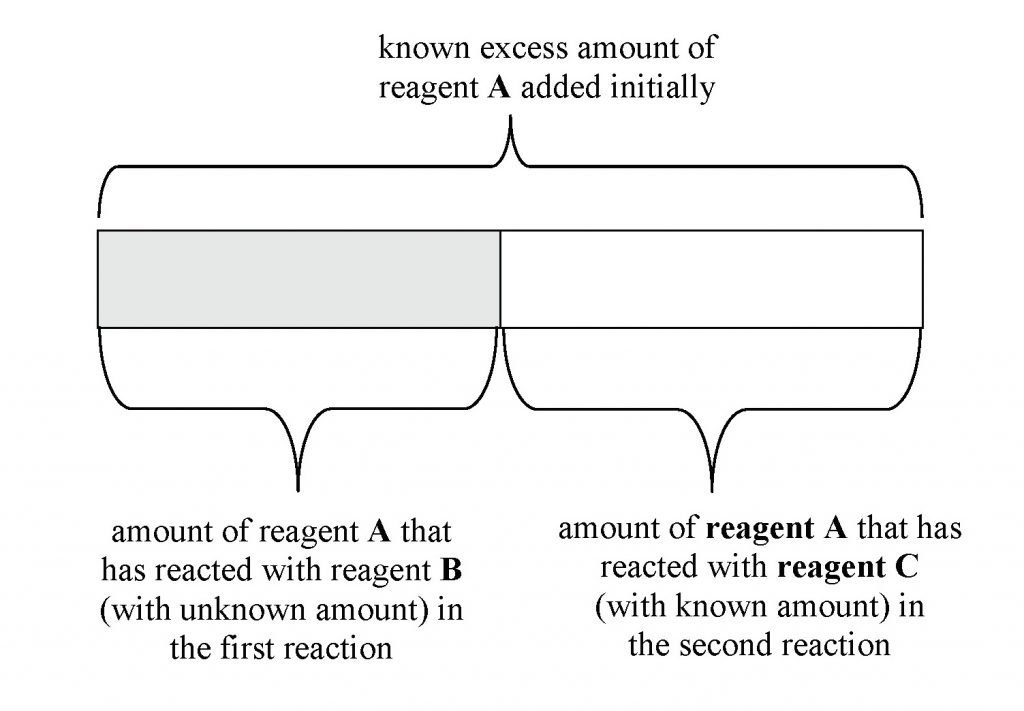
Let’s take a look at an examination question which one of my students in our weekly A-Level JC1 H2 Chemistry Tuition Class asked me recently:
Question:
A sample of impure magnesium was analysed by allowing it to react with excess HCl solution. After 1.32 g of the impure metal was treated with 100 cm3 of 0.750 moldm-3 HCl, the remaining solution required 50 cm3 of a 0.250 moldm-3 aqueous sodium hydroxide for complete neutralisation. Assuming the impurities do not react with the acid, what is the percentage by mass of Mg in the sample? [Ans: 57.5%]
Would you like to take a pen and paper, and try it out on your own first before clicking the video below to look at my suggested answer?
It is important that you master the concepts of Back Titrations because it will appear in both your Chemistry Written Exams as well as your Chemistry Practical Exams!
Click on the following link for Video on 5 Simple Steps to Solve Back Titration Questions in Volumetric Analysis.
H2 Chemistry Video on 5 Simple Steps to Solve Back Titration Questions in Volumetric Analysis
Length of video: 10.53 minutes
Now, if you have come so far in reading my post on Back Titration, how about trying out a Back Titration question on your own? Let me know how you find it. Are you able to solve it easily using the strategies which i have shown in the Chemistry video?
Question:
A sample containing ammonium chloride was warmed with 100 cm3 of 1.00 moldm-3 sodium hydroxide. After all the ammonia had been driven off, the excess sodium hydroxide required 50.00 cm3 of 0.250 moldm-3 sulfuric acid for neutralisation. What mass of ammonium chloride did the sample contain? Leave your answer to 3 significant figures. [Click this link to find the suggested solution]
I hope you find the content easy for your understanding and if you have any questions, leave me a comment below. Feel free to share this blog post with your friends and learn the key H2 Chemistry Concepts together.
Subscribe to my blog to receive 2 updates per month sent to your email!
PS: Under related articles below, there are several blog post discussions and questions related to Back Titrations and Volumetric Analysis. You can also do a keyword search using the search box at the top right hand corner.
Related Articles:
- Tips for JC A-Level H2 Chemistry Paper 4 Practical Exam 2019 – Planning Questions
- Tips for JC A-Level H2 Chemistry Paper 4 Practical Exam 2019
- Ionic Equilibrium: Question on Calculation of Solubility Product, Ksp
- Concentrations of Solutions in Atoms, Molecules & Stoichiometry
- Organic Chemistry – What is Reflux?

Videos of back titration
All year I’ve been trying to understand back titration and you just saved my life😂
Amount of NH4CL is 4.0125g